CITY, Country, 2020-Oct-21 — /EPR Network/ —
Market Report Summary
- Market – Atrial Fibrillation Devices Market
- Market Value – Expand Almost 4X in 2020–2030
- Market CAGR Value – 13% in 2020–2030
- Market Forecast Year – 2020 to 2030
For Full Information -> Click Here
Read Full Press Release Below

Interventional electrophysiology devices have already had a significant impact on patients suffering from cardiac arrhythmias, and are being increasingly adopted as a permanent treatment option for atrial fibrillation. Procedures such as radiofrequency ablation, cryoablation, and cox-maze are well performed in today’s practice.
Recent studies have found cryoablation to be more effective than medical management, and it is widely used in medical surgical practice. The overall market is primarily driven by the growing prevalence of atrial fibrillation amongst the geriatric as well as adult population, and the adoption of minimally-invasive techniques for the permanent treatment of atrial fibrillation.
Moreover, the use of innovative techniques, advancements in technology, and favorable reimbursement scenario are some other factors responsible for propelling the growth of the atrial fibrillation devices market.
Get Sample Copy of Report @ https://www.persistencemarketresearch.com/samples/31531
Companies covered in Atrial Fibrillation Devices Market Report
- Biosensense Webster, Inc. (Johnson & Johnson)
- Abbott Laboratories
- Medtronic Plc
- Boston Scientific Corporation
- AtriCure, Inc.
- Japan Lifeline Co.
- Biotronik, Inc
- MicroPort Scientific Corporation
- Biomerics
- CathRx
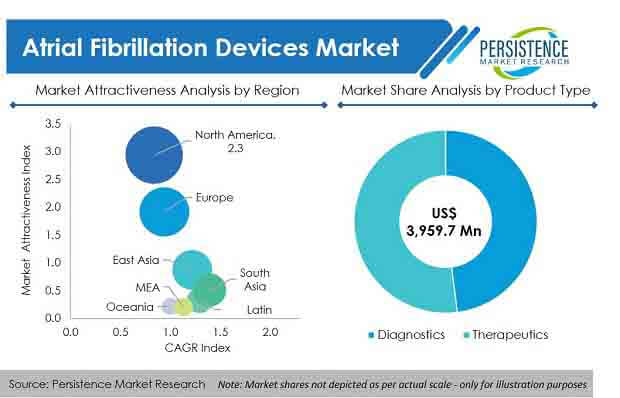
As such, the global atrial fibrillation devices market is estimated to be valued at US$ 3.9 Bn in 2020, with the market expected to exhibit a healthy CAGR of more than 14% during the forecast period (2020-2030).
Key Takeaways from Atrial Fibrillation Devices Market Study
- The diagnostics segment, by product, is expected to gain maximum revenue share over the forecast period. The therapeutic segment is anticipated to increase at a CAGR of 15% over 2020–2030, to reach US$ 8.3 Bn by 2030.
- According to PMR data collected from electrophysiologists in various regions, radiofrequency catheter ablation is the procedure of choice due to the high success rate, while cryoablation is the second preference for atrial fibrillation catheter ablation.
- By technology, the radiofrequency and cryoablation segments, collectively held more than 70% market share in terms of revenue in 2019.
- Among all end users, hospitals is the leading segment, which is followed by cardiac catheterization laboratories. The hospitals segment is anticipated to expand at a CAGR of 14% over the forecast period.
- North America and Europe, collectively held more than 70% revenue share in 2019, while South Asia is expected to show greater growth potential over the forecast period.
- With the ongoing Covid-19 pandemic wreaking havoc across the world, other non-emergency treatments have been deprioritized. As such in the near-term, the growth of the atrial fibrillation devices market will be slower than the average for the decade ahead.
“New product approvals and launches of ablation catheters along with increasing demand for minimally-invasive procedures offering increased longevity and safety profiles are driving the growth of the global atrial fibrillation devices market,” says a PMR analyst.
Access Full Report @ https://www.persistencemarketresearch.com/checkout/31531
New Product Launches – Key Strategic Focus of Market Players
Product approvals by the FDA for new AF devices are responsible for intense competition among market players. Numerous organizations are focusing on new product launches for electrophysiology, especially for atrial fibrillation.
For instance, in August 2016, Biosense Webster, Inc., a world leader in the treatment and diagnosis of heart arrhythmias, announced the launch of THERMOCOOL SMARTTOUCH SF catheters with contact force technology and a porous tip designed to optimize the treatment of atrial fibrillation. Also, in January 2019, Abbott announced FDA approval for the TactiCath Contact Force Ablation Catheter. These winning strategies by leading players in the atrial fibrillation devices market are also being followed by regional and local players.
Atrial Fibrillation Devices Market: Competition Landscape
The global market is set to broaden its scope during the forecast period, stimulated by the rising prevalence of AF, worldwide. In addition, a favorable reimbursement scenario and increasing demand for minimally-invasive procedures have augmented the sales of atrial fibrillation devices.
The market is dominated by 4 major manufacturers – Biosense Webster, Inc. (Johnson & Johnson), Abbott Laboratories, Medtronic Plc, and Boston Scientific Corporation, with a revenue share of over 70% in 2019. These major manufacturers are focusing on manufacturing electrophysiology devices used specifically for atrial fibrillation procedure, such as ablation devices, intracardiac echocardiography catheters, and left atrial appendage (LAA) management devices.
Key market players covered by PMR include Biosense Webster, Inc. (Johnson & Johnson), Abbott Laboratories, Medtronic Plc, Boston Scientific Corporation, ArtiCure, Inc., Japan Lifeline Co., Biotronik SE & Co. KG, Biomerics, MicroPort Corporation, and CathRx, who are consolidating their position through mergers, acquisitions, and new product launches.
Want more insights?
PMR brings a comprehensive research report on forecasted revenue growth at global, regional, and country levels, and provides an analysis of the latest industry trends in each of the sub-segments from 2015 to 2030. The global atrial fibrillation devices market is segmented in detail to cover every aspect of the market and present a complete market intelligence approach to the reader.
The study provides compelling insights on the atrial fibrillation devices market on basis of product (diagnostics – conventional EP catheters, mapping catheters, CS catheters, and ICE catheters) and therapeutics – EP ablation, maze surgery, and LAA management devices), technology (radiofrequency, crythotherapy, ultrasound, and others), and end user (hospitals, ambulatory surgical centers, and cardiac catheterization laboratories), across seven major regions.
Explore Extensive Coverage of PMR`s Life Sciences & Transformational Health Landscape
Persistence Market Research (PMR) is a third-platform research firm. Our research model is a unique collaboration of data analytics and market research methodology to help businesses achieve optimal performance.
To support companies in overcoming complex business challenges, we follow a multi-disciplinary approach. At PMR, we unite various data streams from multi-dimensional sources. By deploying real-time data collection, big data, and customer experience analytics, we deliver business intelligence for organizations of all sizes.
Our client success stories feature a range of clients from Fortune 500 companies to fast-growing startups. PMR’s collaborative environment is committed to building industry-specific solutions by transforming data from multiple streams into a strategic asset.
Contact us:
Ashish Kolte
Persistence Market Research
Address – 305 Broadway, 7th FloorNew York City,
NY 10007 United States
U.S. Ph. – +1-646-568-7751
USA-Canada Toll-free – +1 800-961-0353
Sales – sales@persistencemarketresearch.com
Website – https://www.persistencemarketresearch.com