As per Persistence Market Research’s latest revised industry analysis, the global immunoassay interference blocker market was valued at US$ 223 Mn in 2020, and is expected to exhibit a CAGR of 6.7% over the forecast period (2021-2031).
Substances that modify quantifiable concentration of analytes or change antibody binding can conceivably result in immunoassay interference. Prevalence of interference is usually less in assays holding blocking agents, which counteract or inhibit the interference. Detection of interference may involve usage of a different assay, or quantification before and after treatment with supplementary blocking reagents.
Get Sample Copy of this Report@ https://www.persistencemarketresearch.com/samples/24343
Growing prevalence of chronic diseases and increasing aging population across the world are factors that are expected to surge the need for biochemical testing for the management of chronic diseases. Increase in aging population is likely to escalate demand for diagnostic kits and reagents for the detection of age-related as well as other chronic conditions. This is expected to drive the immunoassay interference blocker market over the forecast period of 2021 to 2031.
Governments around the world are supporting the growth of specialty medical diagnostic reagents, which further drives research & development. Immunoassay interference blocker kits and reagents play a vital role in preventing unnecessary interference generated while preforming an immunoassay-based disease detection test. Hence, an increase in growth of immunoassay interference blockers has been witnessed due to rising government support and funding.
Key manufacturers of immunoassay interference blockers have been focusing on expansion and new product launch programs to increase their consumer base in different geographies.
- In 2019, Bio Rab Laboratories announced the launch of an innovative test to aid in the diagnosis of Lyme disease, with the FDA clearance of the bioplex 2200 Lyme total assay.
- In 2021, Thermofisher Scientific planned for its manufacturing site expansion in the region of Mebane, N.C. USA.
Company Profiles:
- Abcam plc
- AMS BioApplication (Europe) Limited
- Aviva Systems Biology Corporation
- Bio-Rad Laboratories, Inc.
- Meridian Bioscience, Inc.
- Roche Diagnostics GmbH
- Rockland Immunochemicals, Inc.
- Scantibodies Laboratory, Inc.
- Thermo Fisher Scientific
Request for Methodology@ https://www.persistencemarketresearch.com/methodology/24343
Key Takeaways from Market Study
- Antibody interference blockers hold the largest market share as compared to other products, expanding at a CAGR of 6.4%. A similar trend is expected to continue, primarily due to their high efficiency and reliable assay outcomes.
- The application segment is led by antibody capture assays with a share of 39.8% in 2020, owing to high, precise, and accurate results with least interference obtained via immunoassay interference blockers.
- Academic and research institutes dominated the end user segment with a share of around 39.5% in 2020. This segment is expected to grow two-fold by the end of 2031 due to increasing usage of immunoassay interference blockers in research activities to introduce novel applications.
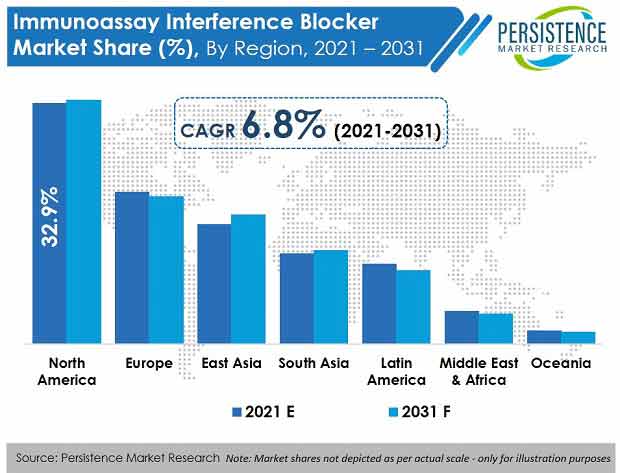
- By region, North America is slated to be the largest regional market with a value share of 33.3% at the end of the forecast period, due to constant clinical efforts to establish advanced diagnostics biologics and new drug discovery in the region.
“Increasing healthcare R&D spending and government support, continual technological advancements, and growing demand for better diagnosis are boosting demand for immunoassay interference blockers,” says an analyst of Persistence Market Research.
Impact of Market Consolidation
Considering consolidation activities in the global market, strategic focus on novel product development and research based on advanced immunoassay interference blockers are some of the key growth strategies by market players. Prominent players are strengthening their product ranges through acquisitions and reaching out to key as well as emerging markets.
For instance:
- In April 2020, Bio Rad Laboratories, Inc. announced the commercial launch of its SARS-CoV-2 Total Ab test, a blood-based immunoassay kit to help determine if an individual has developed antibodies to SARS-CoV-2, the virus associated with COVID-19 disease.
Access Full Report@ https://www.persistencemarketresearch.com/checkout/24343
Market Landscape
The exhaustive research report on global immunoassay interference blocker market has detailed analysis on the various key players involved in this market.
Key companies such as Abcam plc, AMS Biotechnology (Europe) Limited, Aviva Systems Biology Corporation, Bio-Rad Laboratories, Inc., Meridian Bioscience, Inc., Roche Diagnostics GmbH, Rockland Immunochemicals, Inc., Scantibodies Laboratory, Inc. and Thermo Fisher Scientific are profiled in this research study.
What Does the Report Cover?
Persistence Market Research offers a unique perspective and actionable insights on the immunoassay interference blocker market in its latest study, presenting historical demand assessment of 2016 – 2020 and projections for 2021 – 2031.
The research study is based on the product (antibody interference blockers, detection interference blockers, and surface interference blockers), application (sandwich immunoassays, antibody capture assays, competitive ELISA, and others) and end user (biotechnology companies, contract research organizations, academic and research institutes, and molecular diagnostics companies), across seven key regions of the world.
